The Challenge Behind Reactions Using Gas as a Reagent
A major challenge in drug discovery is the high cost of reagents, limited amounts of intermediates available when running a multi-step synthesis, and the pressure (no pun intended) of rapidly evolving project timelines. High-throughput experimentation (HTE) addresses these challenges by enabling rapid optimization of reaction conditions.1 By using HTE, scientists can use less material, reduce catalyst loadings, and evaluate different conditions simultaneously, significantly accelerating the drug discovery process.
One significant challenge in HTE is the use of gases as reagents. This is common in reactions such as the Carbonylation reaction (Image 1), where carbon monoxide is inserted into an organic molecule.2 A Hydrogenation reaction (Image 2), where hydrogen gas, in the presence of a metal catalyst, reduces alkenes and alkynes3, and a Chan-Lam coupling (Image 3), which requires oxygen gas to re-oxidize the copper catalyst.4
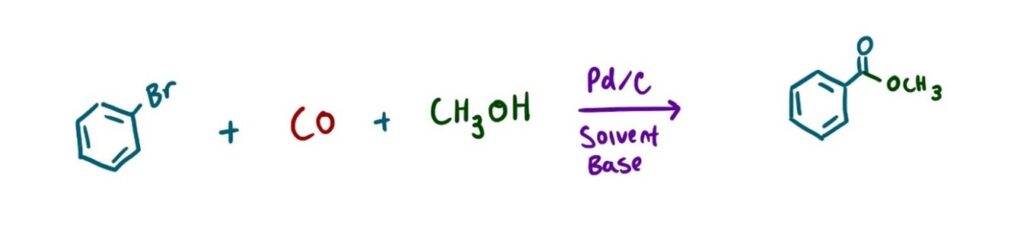
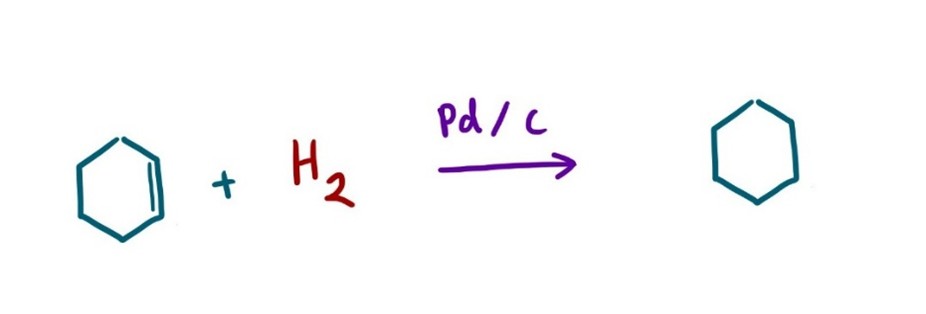
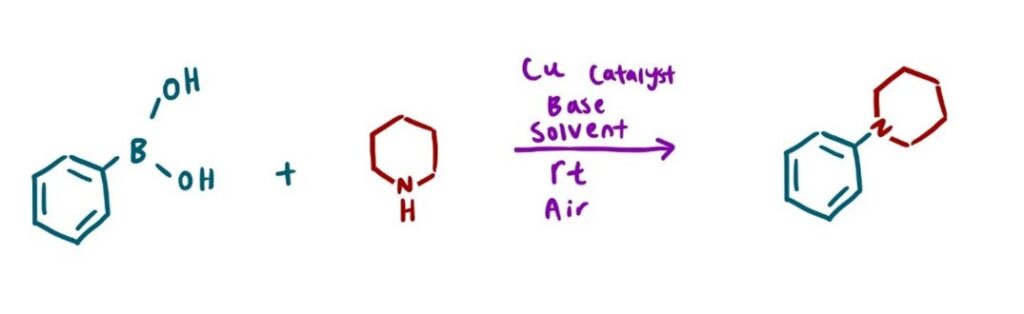
Typically, scientists will use a Parr reactor or a balloon to facilitate gas exchange for these types of reactions. However, these approaches are usually limited to one reaction at a time, making the screening of parallel conditions challenging. Furthermore, adapting these methods to the microscale can be difficult. Filling a balloon with gas is also not ideal for reaction optimization, as a balloon offers little to no control over pressure, limiting the reproducibility and experimental flexibility.
The V&P High Pressure Reactor and its Versatility in Reaction Optimization
The V&P High-Pressure Reactor (HPR) is a versatile tool that enables scientists to test a wide range of conditions for reactions that require gas as a reagent or to provide an inert environment. Available in a 48 or 96-well format, VP 416-ALE-48-HP or VP 416-ALE-96-HP, the HPR works up to 300 psi at 24 °C, and 200 psi at 180 °C. With the HPR at their disposal, scientists can easily screen different solvents, bases, catalysts, ligands, catalyst loadings, and various compounds in parallel at the microscale! This dramatically increases reaction optimization while improving reproducibility and experimental control.
The HPR and Monitoring Pressure During a Reaction
Features such as the integrated pressure gauge on the HPR allow researchers to accurately monitor and control reaction pressure, making it especially useful for determining the minimum pressure necessary without affecting the rate of a reaction. This capability is particularly valuable during reaction optimization, as operating at lower pressures is highly desirable when reactions are scaled up.
The replaceable O-rings are also beneficial for chemists, as many reactions use harsh solvents, which compromise sealing components over time. By replacing the O-rings when needed, researchers can maintain a reliable seal. In addition, the PFA films and rubber mats used for sealing the reactions are disposable, preventing reaction contamination.
Every well inside the HPR can be evenly mixed and heated using the VP 710C5-7A tumble stirrer, and the VP 741D heat block with corresponding adaptor. This allows researchers to study different reaction temperatures and parameters.
Accelerating Drug Development with the HPR
Overall, the HPR is an excellent addition to a scientist’s toolkit. Reaction optimization is a vital component for accelerating the discovery of new compounds, and the HPR enables this by offering precise control, improved efficiency, and faster data generation compared to a traditional batch reaction.
- Nsouli, G. Galiyan, L. K. G. Ackerman-Biegasiewicz, Angew. Chem. Int. Ed.2025, 64, e202506588. https://doi.org/10.1002/anie.202506588
- Haynes, 6.01 – Carbonylation Reactions, Comprehensive Inorganic Chemistry II (Second Edition), Elsevier, 2013, Pages 1-24, ISBN 9780080965291, https://doi.org/10.1016/B978-0-08-097774-4.00601-X.
- Paul Sabatier – Nobel Lecture. NobelPrize.org. Nobel Prize Outreach 2026. Wed. 15 Apr 2026. https://www.nobelprize.org/prizes/chemistry/1912/sabatier/lecture/
- An Efficient Diamine·Copper Complex-Catalyzed Coupling of Arylboronic Acids with Imidazoles
James P. Collman and Min Zhong
Organic Letters 2000 2 (9), 1233-1236
DOI: 10.1021/ol000033j